 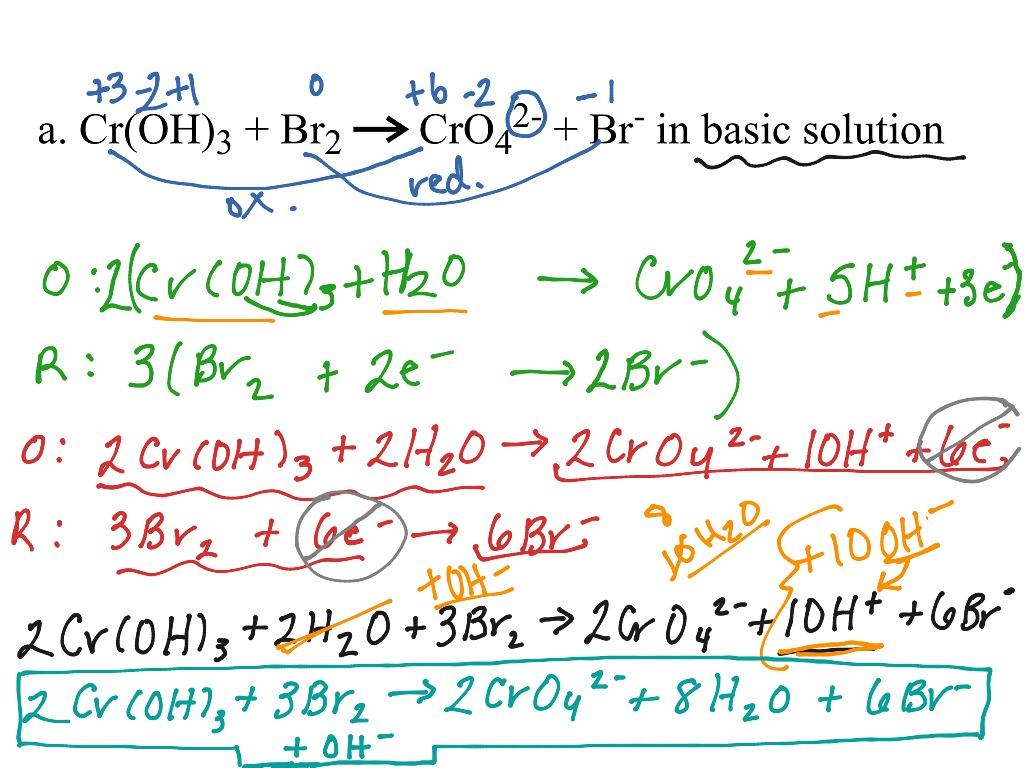
We know that the reaction takes place in an acidic solution. In this case, we will multiply Cr 3+ by 2 in order to balance Chromium atoms. Therefore, we will just balance the reduction part of the reaction. In the example question, the oxidation part of the reaction in terms of Fe atoms is already balanced. In the third step of balancing redox reactions by half-reaction method, we will balance the atoms present in each half of the reaction except O and H atoms. Step 2ĭivide the equation into two separate half reaction-oxidation half and reduction half. Step 1įirst, we have to write the basic ionic form of the equation. The steps to balance this equation is as follows. We have to balance the above redox reaction. The dichromate ions (Cr 2O 7 2–) are reduced to Cr 3+ ions in the reaction. We will demonstrate this method with an example so as to understand the steps of balancing redox reactions by half-reaction method.įor instance, a reaction is given where Fe 2+ ions are converted to Fe 3+ ions by dichromate ions in an acidic solution. Finally, we add them together to give a balanced equation. Thereafter, we balance both the parts of the reaction separately. In this procedure, we split the equation into two halves. Balancing Redox Reactions by Half-Reaction Method We will further understand the steps of balancing redox reactions by solving a problem on the basis of oxidation number method. It will be a balance reaction if there are equal numbers of oxygen atoms present in both the reactant as well as the product the side. Additionally, it is necessary to check the oxygen atoms present in the equation. It is very important to equate the number of hydrogen atoms on each side of the equation by adding water molecules or H 2O molecules. Similarly, if the reaction takes place in the basic solution add OH – ions in the chemical equation. However, if the reaction takes place in acidic solution then add H + ions in the chemical equation. Overall, the ionic charges of reactant and products will be equal. Accordingly, add H + or OH – ions in the appropriate side of the reaction. Keep in mind the involvement of the ions if the reaction occurs in water. This can also mean that the allocation of oxidation numbers is incorrect. This signifies that either the formulas of the reactant or the products are incorrect. If in a case, two substances are either only oxidized or only reduced then this signifies that something is wrong with the chemical reaction. If the numbers are not equal then multiply it to such a number that overall these numbers become equal. Step 3Ĭalculate the oxidation number on the basis of each atom for the given molecule or ion of the chemical reactions. Step 2ĭetermine correctly the atoms that undergo oxidation number change in the given reaction by allocating the oxidation number of the individual elements present in the reaction. The steps of the oxidation number method are as follows: Step 1Ĭorrectly write the formula for the reactants and the products of the chemical reaction. 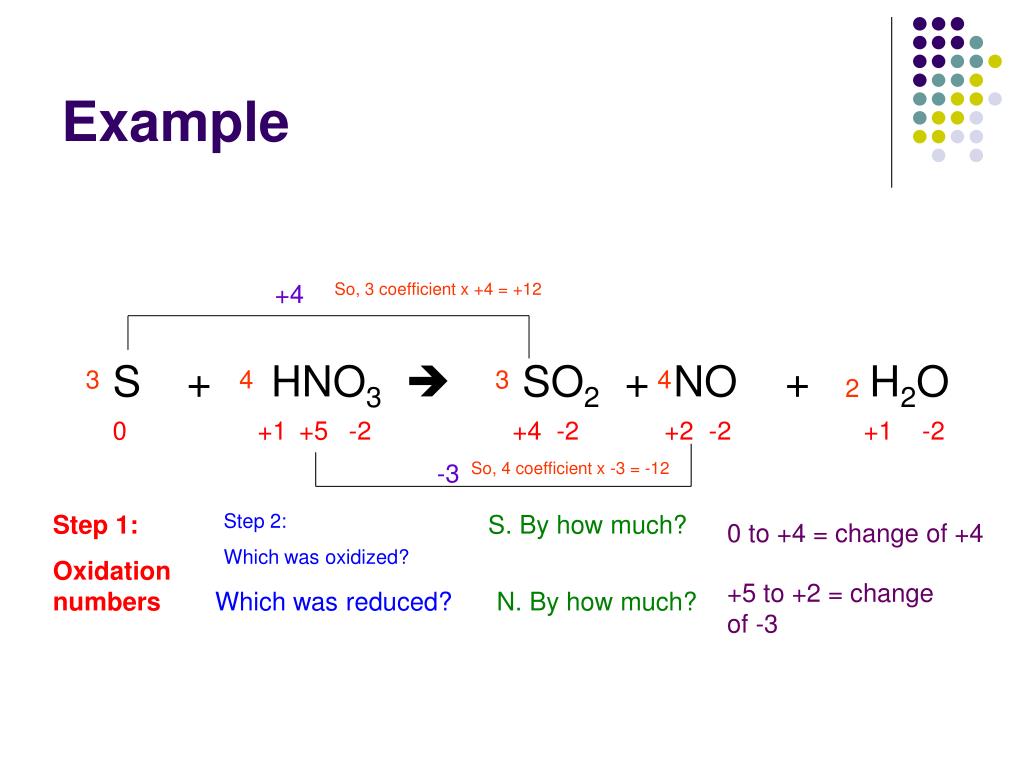
A very important thing to keep in mind while writing oxidation-reduction reactions is to correctly write the compositions and formulas of the substances and products present in the chemical reaction. Balancing Redox Reactions by Oxidation Number MethodĪs with every other reaction, it is very important to write the correct compositions and formulas. It depends on the individual which method to choose and use. Half-reaction method depends on the division of the redox reactions into oxidation half and reduction half. Oxidation number method is based on the difference in oxidation number of oxidizing agent and the reducing agent.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
